Abstract
Background: A novel glycoengineered type II anti-CD20 antibody, MIL62 with a nearly completely afucosylated N-glycans in Fc region, has demonstrated superior activity compared with rituximab and obinutuzumab in vitro and in vivo, respectively. Orelabrutinib (ICP-022) is a novel and highly selective irreversible Bruton's tyrosine kinase (BTK) inhibitor that does not affect IL2-associated tyrosine kinase (ITK) or antibody-dependent cellular cytotoxicity, making it an attractive candidate for combined therapy with anti-CD20 antibodies.
Methods: This was a phase I/IIa dose escalation and dose expansion study (NCT 04304040), which investigated MIL62 combination with orelabrutinib for the treatment of patients with relapsed/refractory (r/r) B-cell non-Hodgkin lymphoma (B-NHL) with received at least one prior systemic regimen. The dose escalation was conducted with a standard 3+3 dose scheme in different dose combinations of MIL62 injection 800 mg or 1000 mg plus orelabrutinib 100 mg or 150 mg oral daily up to 120 weeks or until disease progression, or intolerable toxicity, respectively. The primary endpoint was objective response rate (ORR), defined as a complete response (CR) or partial response (PR) assessed by investigator per Lugano 2014 criteria. The secondary endpoints were duration of response, pharmacokinetics, and safety. Adverse events (AEs) were graded by CTCAE version 5.0.
Results: From July 28th, 2020 to January 26th, 2022, 43 patients enrolled from 10 centers in China received at least one dose of either MIL62 or orelabrutinib. Diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL) and marginal zone lymphoma (MZL) accounted for 72.1% (31), 16.3% (7), 9.3% (4) and 2.3% (1), respectively. Median age was 66.0 (range: 31 to 77) years and 29 (67.5%) patients had ECOG PS score of 1-2. The median number of prior therapies was 2.0 (range: 1 to 7) and 22 (51.2%) patients were primary resistant to rituximab-containing regimens. The median treatment time of MIL62 and orelabrutinib was 4.7 (range: 0.2, 17.5) months and 3.9 (range: 0.2, 14.1) months, respectively.
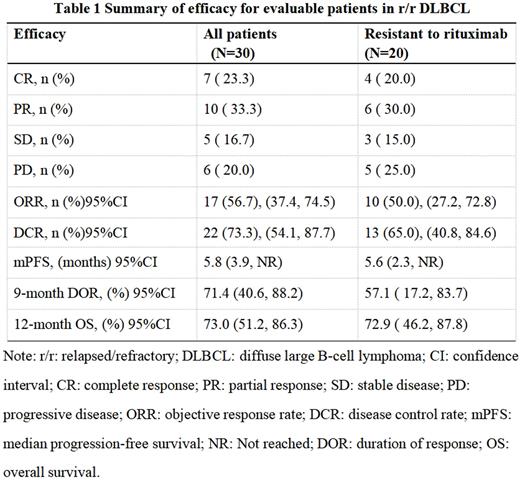
At the cut-off date (July 15th, 2022), the median OS follow-up time was 13.0 (95% confidence interval [95% CI]: 12.2, 15.0) months. Overall, 27 (64.3%) of 42 assessable patients had an objective response, including 13 (31%) with CR and 14 (33.3%) with PR. In addition, among 30 assessable patients with DLBCL, 17 (56.7%) patients had an objective response, including 7 (23.3%) with CR and 10 (33.3%) with PR. The median progression-free survival (PFS), 9-month remission rate and 12-month overall survival rate were 5.8 ([95%CI]: 3.9, Not reached) months, 71.4%, 73.0%, respectively. Especially, in 20 patients with resistance to rituximab, 10 (50%) patients had an objective response, and median PFS was 5.6 months (Table 1).
Among 43 safety-evaluable patients, treatment emergent adverse events (TEAEs) and treatment-related adverse events (TRAEs) occurred in 39 (90.7%) patients, 38 (88.4%) patients respectively. Grade 3 or above TEAE were observed in 20 (46.5%) patients, among which TRAEs in 16 (37.2%) patients, with neutropenia (20.9%), thrombocytopenia (16.3%). Severe adverse events (SAEs) occurred in 14 (32.6%) patients, among which 11 (25.6%) were TRAEs. 4 (9.3%) patients were permanently stopped the study due to TEAEs. The incidence of MIL62 infusion-related reactions was 11.6%, all of which were grade 1-2.
Conclusion: MIL62 combined with orelabrutinib showed promising efficacy in previously treated patients with r/r DLBCL, including those with resistance to rituximab, and has a manageable safety profile.
Keywords: MIL62; type II anti-CD20 monoclonal antibody; r/r B-NHL; DLBCL;
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal